Levocabastine
Consumer Medicine Information
What is in this leaflet
This leaflet answers some common questions about LIVOSTIN. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you using LIVOSTIN against the benefits this medicine is expected to have for you.
If you have any concerns about using LIVOSTIN, ask your doctor or pharmacist.
Keep this leaflet with your medicine. You may need to read it again.
What LIVOSTIN is used for
LIVOSTIN eye drops are used to relieve watery, itchy, swollen eyelids or red eyes caused by allergies to grass, pollen, moulds, dust or other substances.
LIVOSTIN nasal spray is used to relieve sneezing, runny nose or irritation of the nose caused by allergies to grass, pollen, moulds, dust or other substances.
LIVOSTIN contains levocabastine which belongs to a group of medicine called antihistamines. It provides rapid and long lasting relief for symptoms of allergy. It works by blocking the effect of histamine which is a chemical released by the body that causes most of the symptoms.
Your doctor may have prescribed LIVOSTIN for another use. Ask your doctor if you have any questions about why this medicine has been prescribed for you.
Before you use LIVOSTIN
When you must not use it
Do not use LIVOSTIN if
- you have an allergy to LIVOSTIN or any of the ingredients. See Product description at the end of this leaflet for a list of ingredients.
- if the packaging is torn or shows signs of tampering
- beyond the expiry date (month and year) printed on the pack.
Before you start to use it
You must tell your doctor if
- you are pregnant or planning to become pregnant.
- you are breast feeding or wish to breast feed.
- you have or have ever had kidney disease.
- you wear soft contact lenses. As with all eye drops containing benzalkonium chloride, you are advised not to wear soft contact lenses while being treated with LIVOSTIN eye drops.
There is limited experience of use of LIVOSTIN in children under 6 years of age. Ask your doctor for further information if you have any questions on the use of this medicine in children.
If you have not told your doctor or pharmacist about any of the above, tell them before you start using LIVOSTIN.
Your doctor will advise you whether or not to use LIVOSTIN or if you need to adjust the dose or adapt your treatment.
Taking other medicines
Tell your doctor or pharmacist if you are taking other medicines, including medicines you can buy without a prescription from a pharmacy, supermarket or health food shop.
The decongestant oxymetazoline for example may reduce the absorption of nasal levocabastine.
Effects on driving and operating machinery
- LIVOSTIN does not usually affect alertness or concentration. However, should you feel drowsy be careful while driving or operating machinery.
Effects of alcohol
- At the usual dose, LIVOSTIN nasal spray does not usually increase the effect of alcohol.
Using LIVOSTIN
How much to use
Eye drops –
Adults and children 6 years of age and over: the usual dose is 1 drop of LIVOSTIN eye drops in each eye, twice daily. If necessary, the dose may be increased to 1 drop 3 or 4 times daily.
Do not use for more than 8 weeks.
Nasal spray –
Adults and children 6 years of age and over: the usual dose is 2 sprays of LIVOSTIN nasal spray in each nostril, twice daily. If necessary, the dose may be increased to 2 sprays 3 or 4 times daily.
Do not use for more than 8 weeks.
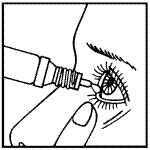
How to use LIVOSTIN eye drops
You may find it easier to put eye drops in your eye while you are sitting or lying down.
- Shake the bottle well before removing the cap. To open a new bottle, twist off the plastic protective cap and the seal will break.
- Hold the bottle upside down in one hand between your thumb and index finger. Using your other hand, gently pull down your lower eyelid to form a pouch.
- Tilt your head back and look up.
- Put the tip of the bottle close to your eye. Do not let it touch your eye.
- Release one drop onto your eye by gently squeezing the bottle. Close your eye. Do not blink or rub your eye.
- While your eye is closed, apply gentle pressure for a few minutes with your fingers to the bridge of your nose to prevent medication from being drained from the eye.
- Repeat the above steps for the other eye.
- Place the cap on the bottle and close it tightly.
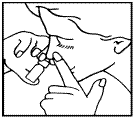
How to use LIVOSTIN nasal spray
Place your index and middle fingers on the white collar of the bottle. Hold the bottle upright by placing your thumb at the bottom of the bottle.
For each new bottle of LIVOSTIN nasal spray, remove the cap and press the white collar of the spray downwards once or twice until a fine spray appears in the air.
- Gently blow your nose to clear the nostrils.
- Shake the bottle well and remove the cap.
- Lean your head slightly forward. Keep one nostril tightly closed. Insert the nozzle into the other nostril.
- Press the white collar down to release each spray. Breathe in through the same nostril between each spray.
- Repeat steps 3 and 4 for the other nostril.
- After use, wipe the nozzle with a tissue and replace the cap.
If you do not understand the instructions provided with this medicine, ask your doctor or pharmacist for help.
If you forget to use it
- Use your dose as soon as you remember, and then continue to use it as you would normally.
- If it is almost time for your next dose, skip the dose you missed and use your next dose when you are meant to.
- Do not use a double dose to make up for the one you missed.
If you have missed more than one dose, or are not sure what to do, check with your doctor or pharmacist.
If you have trouble remembering when to use your medicine, ask your pharmacist for some hints.
If you have used too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre for advice, or go to Accident and Emergency at your nearest hospital.
Do this even if there are no signs of discomfort or poisoning. You may need medical attention.
Poisons Information Centre telephone numbers:
- Australia: 13 11 26
- New Zealand: 0800 764 766
Keep these telephone numbers handy.
If, by accident, you use or drink a whole bottle of LIVOSTIN, you may feel drowsy.
In the meantime, the best thing to do is to drink a lot of water.
While you are using LIVOSTIN
Things you must do
- Always follow your doctor’s instructions carefully.
- Tell your doctor if you become pregnant while using LIVOSTIN.
- If you are about to start taking a new medicine, tell your doctor and pharmacist that you are using LIVOSTIN.
Things you must not do
- Do not use LIVOSTIN to treat any other complaint unless your doctor says so.
- Do not give this medicine to anyone else, even if their symptoms seem similar to yours.
Side effects
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some side effects. Do not be alarmed by this list of possible side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if you experience any of the following:
- slight irritation to your eyes e.g. eye pain, itching, burning or redness of the eyes, conjunctivitis, swelling of the eye, inflammation of the eyelid, watery eyes
- congestion of blood in the eye
- slight irritation to your nose e.g. nasal congestion, nasal discomfort, pain, burning or dryness
- headache
- drowsiness
- nose bleeds
- blurred vision
- hives
- allergic reaction (swelling of the lips, eyelids and tongue, shortness of breath)
- nausea
- cough
- dizziness
- fatigue
- throat pain
- achiness
- difficulty breathing
- abnormally fast heartbeats
- skin irritation
- hypersensitivity
- a general feeling of being unwell
Rarely, allergic reactions may occur. Should this happen discuss the matter with your doctor or pharmacist.
Other side effects not listed above may also occur in some people. Tell your doctor if you notice any other effects.
After using LIVOSTIN
Storage
- Keep LIVOSTIN in a cool, dry place where the temperature stays below 25°C.
- Do not store LIVOSTIN or any medicines in the bathroom or near a sink. Do not leave medicines in the car or on window sills. Heat and dampness can destroy some medicines.
- Keep your medicines where young children cannot reach them. A locked cupboard at least one-and-a-half metres (1.5m) above the ground is a good place to store medicines.
Disposal
Discard LIVOSTIN eye drops one month after the date of first opening. If your doctor tells you to stop using LIVOSTIN, or your medicine has passed its expiry date, ask your pharmacist what to do with any medicine which may be left over.
Product description
What it looks like
LIVOSTIN eye drops contains 4 mL of liquid in a 5 mL volume plastic bottle. LIVOSTIN nasal spray contains 10 mL of liquid in a 15 mL volume plastic bottle.
Ingredients
LIVOSTIN
- contains levocabastine 0.5 milligrams per millilitre.
- The eye drops also contain propylene glycol, polysorbate 80, sodium phosphate dibasic, sodium phosphate monobasic, hypromellose, water and benzalkonium chloride, disodium edetate as preservatives.
- The nasal spray also contains propylene glycol, polysorbate 80, sodium phosphate dibasic, sodium phosphate monobasic, hypromellose, water and benzalkonium chloride, disodium edetate as preservatives.
Sponsor
Johnson & Johnson Pacific
45 Jones Street,
Ultimo NSW 2007
AUSTRALIA
and
Auckland,
NEW ZEALAND
For further information call our toll free info lines:
AUS: 1800 029 979
NZ: 0800 446 147
This leaflet was prepared in December 2014.
Published by MIMS June 2016
