Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using ANDROFEME 1?
ANDROFEME 1 contains the active ingredient testosterone. ANDROFEME 1 is used to treat postmenopausal women with hypoactive sexual desire dysfunction. For more information, see Section 1. Why am I using ANDROFEME 1? in the full CMI.
2. What should I know before I use ANDROFEME 1?
Do not use if you have ever had an allergic reaction to testosterone, tree nuts (almond oil) or any of the ingredients listed at the end of the CMI. Do not use if you have or have had any of the following:
- breast cancer or any other cancer which your doctor has described as being caused or stimulated by androgen
- kidney disease known as nephrotic syndrome
- blockage of a blood vessel by a blood clot (thromboembolism)
- high calcium levels in the blood (hypercalcaemia)
Do not use if you: still menstruate; are pregnant or breastfeeding; are a child.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding. For more information, see Section 2. What should I know before I use ANDROFEME 1? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with ANDROFEME 1 and affect how it works. A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use ANDROFEME 1?
- The usual starting dose is 0.5 mL (5 mg) of cream by measured applicator per day.
- Detailed instructions on how to safely use the applicator syringe and cream can be found in Section 4. How do I use ANDROFEME 1? in the full CMI.
5. What should I know while using ANDROFEME 1?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using ANDROFEME 1? in the full CMI.
6. Are there any side effects?
Common side effects include: acne and oily skin; increased body hair particularly on the face; loss of head hair (male pattern baldness) or thinning; headache; abdominal bloating; constipation.
Serious side effects include: symptoms of allergic reaction; nausea and vomiting; yellowing of the skin and/or eyes; swelling of the ankles; weight gain; persistent headaches; deepening of the voice; changes in tissue of the breast; vaginal bleeding; ovulation and menstrual periods may stop in premenopausal women; enlargement of the clitoris.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
ANDROFEME® 1(and-ro-feme)
Active ingredient: testosterone
Consumer Medicine Information (CMI)
This leaflet provides important information about using ANDROFEME 1. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using ANDROFEME 1.
Where to find information in this leaflet:
1. Why am I using ANDROFEME 1?
2. What should I know before I use ANDROFEME 1?
3. What if I am taking other medicines?
4. How do I use ANDROFEME 1?
5. What should I know while using ANDROFEME 1?
6. Are there any side effects?
7. Product details
1. Why am I using ANDROFEME 1?
ANDROFEME 1 contains the active ingredient testosterone. Testosterone belongs to a group of medicines called androgens and this form of testosterone is identical to the testosterone produced by the ovaries of women and the testes of men.
ANDROFEME 1 is used to treat hypoactive sexual desire dysfunction (HSDD) in postmenopausal women.
Women with this condition often feel distress because they have a very low sexual desire/interest to have sex.
Testosterone can help reduce sexual concerns and distress in postmenopausal women, by improving:
- sexual desire
- pleasure
- orgasm
- responsiveness
- self-image
ANDROFEME 1 is a cream you apply to your skin. It is a simple and effective way of getting testosterone into your body. Your skin absorbs testosterone, which then travels into your bloodstream and throughout your body.
2. What should I know before I use ANDROFEME 1?
Warnings
Do not use ANDROFEME 1 if:
- you are allergic to testosterone, tree nuts (almond oil) or any of the ingredients listed at the end of this leaflet.
- Always check the ingredients to make sure you can use this medicine.
- you have or have had any of the following:
– breast cancer
– any other cancer which your doctor has described as being caused or stimulated by androgen
– kidney disease known as nephrotic syndrome
– blockage of a blood vessel by a blood clot (thromboembolism)
– high calcium levels in the blood (hypercalcaemia) - you still menstruate
- you are pregnant or breastfeeding
- you are a child
Check with your doctor if you:
- have any other medical conditions, especially:
– allergies to any other medicines, foods, preservatives or dyes
– heart disease or high blood pressure
– liver disease
– kidney disease. - take any medicines for any other condition
- are a high level athlete. You need to be aware of the rules governing testosterone (an androgen) use if prescribed ANDROFEME 1 cream as testosterone may enhance athletic performance.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
- Do not use this medicine if you are pregnant or breastfeeding.
It may affect your developing baby if you take it during pregnancy.
The active ingredient in ANDROFEME 1 passes into breast milk and there is a possibility that your baby may be affected.
ANDROFEME 1 should only be used by women who no longer have their menstrual periods, either naturally or because they have had a total hysterectomy.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with ANDROFEME 1 and affect how it works. These include:
- oral oestrogens, medicines contained in oral contraceptives and hormone replacement therapy
- tibolone, a medicine used for hormone replacement
- oral anticoagulants, medicines used to thin blood and to treat or prevent blood clots
- corticosteroids or adrenocorticotropic hormone (ACTH), hormones used to control inflammation
- oxyphenbutazone, a medicine used to treat pain and inflammation
- insulin, a medicine used to control blood sugar levels
- bupropion, a medicine used to aid in smoking cessation
- ciclosporin, a medicine used to suppress the immune system.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect ANDROFEME 1.
4. How do I use ANDROFEME 1?
How much to use
- Follow the instructions provided and use ANDROFEME 1 until your doctor tells you to stop.
- The usual starting dose is 0.5 mL (5 mg) of cream by measured applicator per day.
- You should not use more than 1.0 mL (10 mg testosterone) of cream per day unless directed to do so by your doctor.
- If you take the wrong dose, ANDROFEME 1 may not work as well and your problem may not improve.
- You will need to return to your doctor three to six weeks after starting treatment so that the dose can be checked via a blood test and adjusted if required.
- You should ideally attend the same laboratory for blood testing.
- It may take 1-2 months for you to notice an improvement. If symptoms do not improve after 6 months, you should discuss an alternative treatment with your doctor.
- Continue taking your medicine for as long as your doctor tells you.
- ANDROFEME 1 is usually used for a minimum of 3 months to see if the medicine works for you.
- There is currently limited safety information on the use of ANDROFEME 1 beyond two years. Your doctor will discuss the benefits and risks of longer use with you.
When to use ANDROFEME 1
- Apply ANDROFEME 1 once per day at approximately the same time each day.
- Applying the cream at the same time each day will have the best effect. It will also help you remember when to apply it.
- Avoid swimming or showering until at least four hours after application of ANDROFEME 1 cream.
How to use ANDROFEME 1
- To open the tube, remove the cap and peel off the foil seal.
- A measuring applicator (syringe style) in a sealed sleeve is enclosed in the ANDROFEME 1 box. The applicator is marked with 0.5 mL graduations for dosing accuracy.
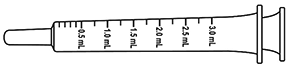
- To measure the correct dose of cream:
– gently squeeze the base of the ANDROFEME 1 tube until cream reaches the open nozzle of the tube.
– insert the tip of the applicator into the open nozzle of ANDROFEME 1 cream so that the nozzle and the shoulder of the applicator are in contact.
– invert the tube and the applicator so the cream will flow with gravity when squeezed.
– carefully squeeze the base of the ANDROFEME 1 tube and at the same time slowly withdraw the red plunger of the applicator. The cream will flow into the barrel of the applicator. Do not try to squeeze the cream into the syringe.
– fill the applicator to the required dose. For example: a 0.5 mL dose of ANDROFEME 1 (5 mg testosterone) needs the flat part of the plunger level with the 0.5 mL mark.
– if there are any air bubbles in the measured dose, fill slightly past the required dose mark then depress the plunger so that the excess cream flows back into the tube. Stop at the required dose mark.
– remove the applicator from the nozzle of the tube and replace the cap firmly on the tube. - To apply the cream:
– always apply ANDROFEME 1 directly onto clean, dry, healthy skin of either the upper outer thigh or buttock
– massage the cream into the area until absorbed. Typically, this takes about 30 seconds.
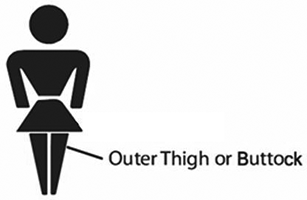
– avoid using perfume, deodorant or moisturising creams/gels on the area because this may interfere with ANDROFEME 1 being absorbed
– cover the application area with clothing once applied
– never apply the cream to broken or damaged skin. Do not use ANDROFEME 1 on the genital or other areas.
- Following application:
– wash your hands thoroughly with soap and water after applying the cream.
– be careful to avoid potential transfer to pregnant women.
– rinse the applicator in hot water after use and replace in the box with the ANDROFEME 1 cream. - For contacts:
– If someone you know comes into regular contact with the skin where you have applied the cream, advise them to wash the affected area on their body with warm, soapy water as soon as possible.
If you forget to use ANDROFEME 1
ANDROFEME 1 should be used regularly at the same time each day. If you miss your dose at the usual time, apply your dose as soon as you remember and continue to apply it as you would normally.
If it is almost time for your next dose, skip the dose you missed and apply your next dose when you are meant to.
Do not apply a double dose to make up for the dose you missed.
If you use too much ANDROFEME 1
If you think that you have used too much ANDROFEME 1, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre (by calling 13 11 26) for advice, or
- contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using ANDROFEME 1?
Things you must do
- If you become pregnant while taking this medicine, tell your doctor immediately.
- If you are about to have any blood tests, tell your doctor that you are taking this medicine. It may interfere with the results of some tests.
- Keep all of your doctor’s appointments so that your progress can be checked.
Your doctor may do some blood tests from time to time to make sure the medicine is working and to prevent unwanted side effects. - For women over 50 years it is recommended that a mammogram is performed every 2 years, unless otherwise advised by your doctor.
Call your doctor straight away if you:
- see any signs of increased levels of testosterone in a child or partner that may have occurred through accidental exposure to ANDROFEME 1.
ANDROFEME 1 may be transferred to another person during close skin contact with the area where the cream has been applied.
If close contact occurs repeatedly or for long periods of time, especially with children, this could cause signs of increased testosterone (virilisation). Signs of increased testosterone in children include:
- enlarged penis or clitoris
- early development of pubic hair
- increased erections or sex drive
- aggressive behaviour.
Avoid unwanted transfer to another by wearing clothes which cover the application area and/or by showering before contact. You need to ensure that your partner is not put at risk of receiving testosterone by accident.
Remind any doctor, dentist or pharmacist you visit that you are using ANDROFEME 1.
Things you should not do
- Do not take ANDROFEME 1 to treat any other complaints unless your doctor tells you to.
- Do not give your medicine to anyone else, even if they have the same condition as you.
- Do not stop using ANDROFEME 1, or change the dosage, without checking first with your doctor. If you stop using it suddenly, your condition may worsen, or you may have unwanted side effects.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how ANDROFEME 1 affects you.
Looking after your medicine
Follow the instructions in the carton on how to take care of your medicine properly.
- Keep the tube sealed until time for use. Replace cap firmly on the tube once opened.
- Do not transfer the cream out of the original tube into another container.
- Rinse the applicator in hot water after use and replace in box with ANDROFEME 1 cream ready for the next day’s application.
Store it in a cool dry place where the temperature stays below 25°C. Store away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Do not freeze.
Keep it where young children cannot reach it.
When to discard your medicine
Once opened, the contents of the tube should be used within 125 days.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Hormone-related:
Stomach-related:
|
Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Hormone-related:
Allergic reaction-related:
|
Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor’s prescription.
What ANDROFEME 1 contains
| Active ingredient (main ingredient) |
Testosterone – 10 mg testosterone per 1 mL of cream |
| Other ingredients (inactive ingredients) |
butylated hydroxytoluene carbomer 940 cetomacrogol 1000 cetostearyl alcohol citric acid dl-alpha-tocopherol acetate phenoxyethanol purified water trolamine |
| Potential allergens | almond oil butyl hydroxybenzoate ethyl hydroxybenzoate isobutyl hydroxybenzoate methyl hydroxybenzoate propyl hydroxybenzoate |
Do not take this medicine if you are allergic to any of these ingredients.
What ANDROFEME 1 looks like
ANDROFEME 1 is a white, opaque, odourless cream in a sealed laminated 50 mL tube.
The crimp at the base of the tube has the batch number and expiry date imprinted on it.
ANDROFEME 1 is supplied as a 50 mL sealed tube with a dose applicator marked with 0.25 mL graduations in a carton.
(AUST R 324274).
Who distributes ANDROFEME 1
Lawley Pharmaceuticals Pty Ltd
Unit 2/15A Harrogate Street
West Leederville WA 6007
Telephone: +61 8 9388 0096
Toll-Free phone (Australia): 1800 627 506
Email: info@lawleypharm.com.au
This leaflet was prepared in September 2023.
Published by MIMS October 2023




